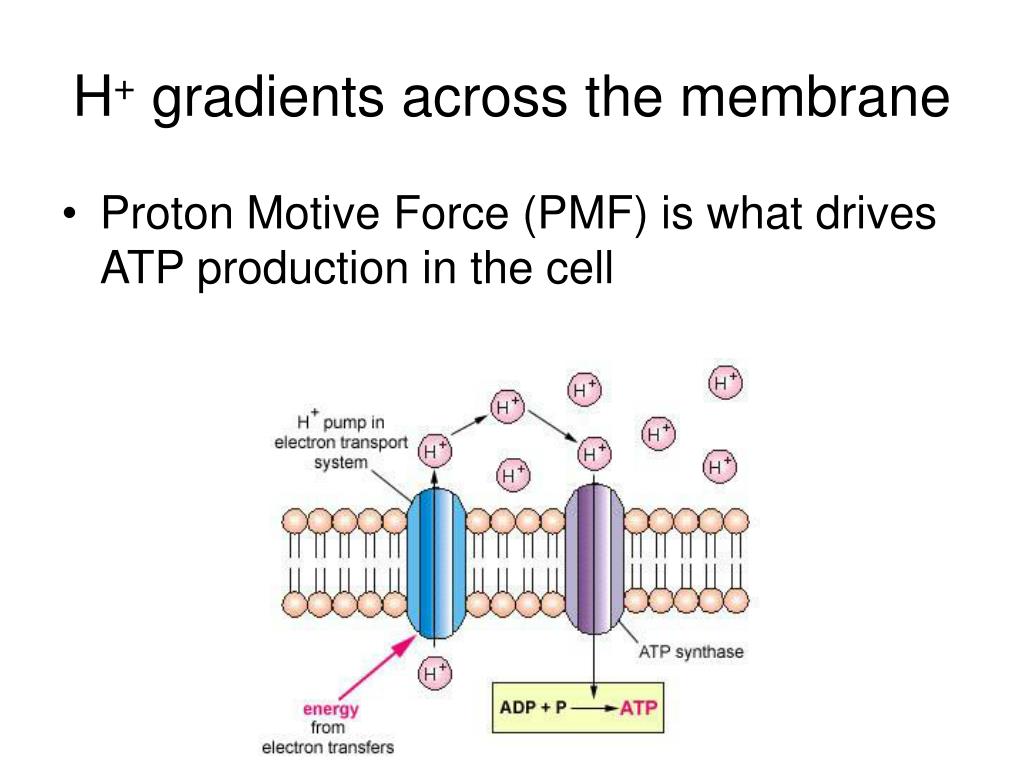
62, p165-169 (2022).ĪTPase bacterial flagella flagellar assembly protein export proton motive force transmembrane ion channel type III secretion system.Īctivation mechanism of the dual-fuel export engine in the fT3SS. This review article is an extended version of a Japanese article, Membrane voltage-dependent activation of the transmembrane export gate complex in the bacterial flagellar type III secretion system, published in SEIBUTSU BUTSURI Vol. In this article, we review our current understanding of the activation mechanism of the dual-fuel protein export engine of the fT3SS. Furthermore, the export gate complex also has a sodium ion (Na +) channel to couple Na + influx with flagellar protein export. However, when the electric potential difference, which is defined as membrane voltage, rises above a certain threshold value, the export gate complex becomes an active H +/protein antiporter to a considerable degree, suggesting that the export gate complex has a voltage-gated activation mechanism. When the ATPase ring complex does not work well in a given environment, the export gate complex will remain inactive. 
ATP hydrolysis by the cytoplasmic ATPase complex converts the export gate complex into a highly efficient proton (H +)/protein antiporter that couples inward-directed H + flow with outward-directed protein export. The transmembrane export gate complex is fueled by proton motive force across the cytoplasmic membrane and is divided into four distinct functional parts: a dual-fuel export engine a polypeptide channel a membrane voltage sensor and a docking platform. The fT3SS of Salmonella enterica serovar Typhimurium is composed of a transmembrane export gate complex and a cytoplasmic ATPase ring complex. Bacteria employ the flagellar type III secretion system (fT3SS) to construct flagellum, which acts as a supramolecular motility machine.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
